Sample information |
|
| Picture |

|
|---|---|
| Location | |
| Collection date | 02/01/2025 |
| Captive / Cultivated? | Wild-caught |
| Group | Berkshire Community College |
| Observations | Spider was collected from the top left-hand corner of a sliding door in the back side of the house. |
| Putative identification | Arthropoda Arachnida Araneae Theridiidae |
Methods |
|
| Extraction kit | Monarch DNA extraction (NEB) |
| DNA extraction location | Abdomen |
| Single or Duplex PCR | Duplex Reaction |
| Gel electrophoresis system | Edvotek Gel Electrophoresis |
| Buffer | TAE |
| DNA stain | SYBR Safe |
| Gel images |

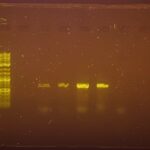
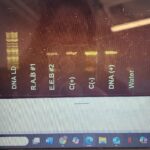
|
| Protocol notes | Taq polymerase used: New England Biolabs One Taq hot Start Quick-Load 2x MM w/Standard Buffer (product # M0488S) DNA Ladder Used: New England Biolabs 1 kb Plus DNA Ladder for Safe Stains (product #N0559S) First single PCR ran on 3/04/2025 at an annealing temperature of 49 degrees Celsius with arthropod primers. The results were inconclusive as the Theridiidae sample did not present with any band but the controls did provide distinct bands as well as the Steatoda Triangulosa sample. No signs of contamination. Second Duplex PCR was ran on 4/1/2025 at an annealing temperature of 49 degrees Celsius with both arthropod and Wolbachia primers. Two bands were distinctively seen for each control sample at 400 and another above each at 700. Contamination was evident as the water control had a distinct band as well. |
Results |
|
| Wolbachia presence | Unknown |
| Confidence level | Low |
| Explanation of confidence level | My confidence level is based on the previous Single PCR reaction that did not show any DNA for the arthropod primer RAB#1 but did have a positive reaction with all other primers. This time we are using a Duplex PCR with both arthropod and Wolbachia primers. |
| Wolbachia 16S sequence | |
| Arthropod COI sequence |
|
| Summary | |
 Umbrella Wasp
Umbrella Wasp Fannia Canicularis
Fannia Canicularis Oulema Obsucra JM2
Oulema Obsucra JM2 Collembola
Collembola Linyphiidae BK1
Linyphiidae BK1