Sample information |
|
| Picture |


|
|---|---|
| Location | |
| Collection date | 12/01/2025 |
| Captive / Cultivated? | Wild-caught |
| Group | Coalinga High School |
| Observations | I found this spider crawling on the floor inside of my house. The temperature of my house was probably around 70 degrees Fahrenheit. It was crawling out in the open, and I noticed that it was pretty large. It was fuzzy, and it had a few black spots, but it mostly had spots in different shades of brown. It also had long legs and looked fairly thick for a spider. |
| Putative identification | Arthropoda Arachnida Araneae |
Methods |
|
| Extraction kit | DNeasy (Qiagen) |
| DNA extraction location | Partial abdomen |
| Single or Duplex PCR | Single Reaction |
| Gel electrophoresis system | MiniOne |
| Buffer | TBE |
| DNA stain | GelGreen |
| Gel images |

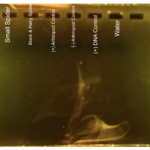
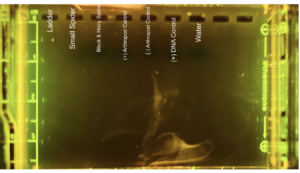
|
| Protocol notes | I followed MiniOne protocol. DNA Extraction: I know that we had some cross-contamination in the positive arthropod control during DNA extraction. When we were pipetting the different reagents into our controls, we forgot to change the tip before pipetting into our positive arthropod control, causing possible cross-contamination. Arthropod Gel Electrophoresis lanes: 1- 100bp ladder 2 – The second well is our small spider. 3 – The third well is our black and hairy spider. 4 – We accidentally skipped this well, so there is nothing in this well. 5 – The fifth well is our positive arthropod control. 6 – The sixth well is our negative arthropod control. 7 – The seventh well is our positive DNA control. 8 – The eight well is our water Analysis: The controls worked for the arthropod electrophoresis, but we cannot be confident about the positive arthropod control, due to cross-contamination that took place. None of our samples had bands, meaning that it is possible for us to not have pr homogenized the samples correctly.
Loading the Gel: When we loaded the ladder, I believe that we may have improperly loaded it because we had what seemed like a faint and very smudged ladder that you cannot make anything of. Wolbachia Gel Electrophoresis Lanes: 1 – 100bp ladder 2- The second well is our small spider. 3 – The third well is our black and hairy spider. 4 – The fourth well is our positive arthropod control. 5 – The fifth well is our negative arthropod control. 6 – The sixth well is our positive DNA control. 7 – The seventh well is our water. Analysis: We only received one very faint band for the positive DNA control. I think this was because of improper loading. There is a part of the gel at the bottom that shows a hazy and smeared liquid, which could be the result of the improper loading.
|
Results |
|
| Wolbachia presence | Unknown |
| Confidence level | Low |
| Explanation of confidence level | I’m not very confident about our results because something went wrong while loading the ladder for the Wolbachia Gel Electrophoresis. In the arthropod gel electrophoresis, we did not receive bands for either of our samples, which could be the result of not properly grinding the arthropods, meaning no DNA was released, so we were unable to have any results. I think that the improper loading of the ladder played a part to not having any bands, but I think that even if the ladder was properly loaded, we wouldn’t have had any results since we had no bands for the arthropod gel. |
| Wolbachia 16S sequence | |
| Arthropod COI sequence |
|
| Summary | |

 BRI-MAO COLEOPTERA
BRI-MAO COLEOPTERA BRI-AMM Hemiptera
BRI-AMM Hemiptera BRI-ANH Order Diptera
BRI-ANH Order Diptera BORPARHemiptera
BORPARHemiptera BRI-KMT Hymenoptera
BRI-KMT Hymenoptera